What calcification means, how to check if you’re affected, and what to do next.
Published by Justpoint Inc.
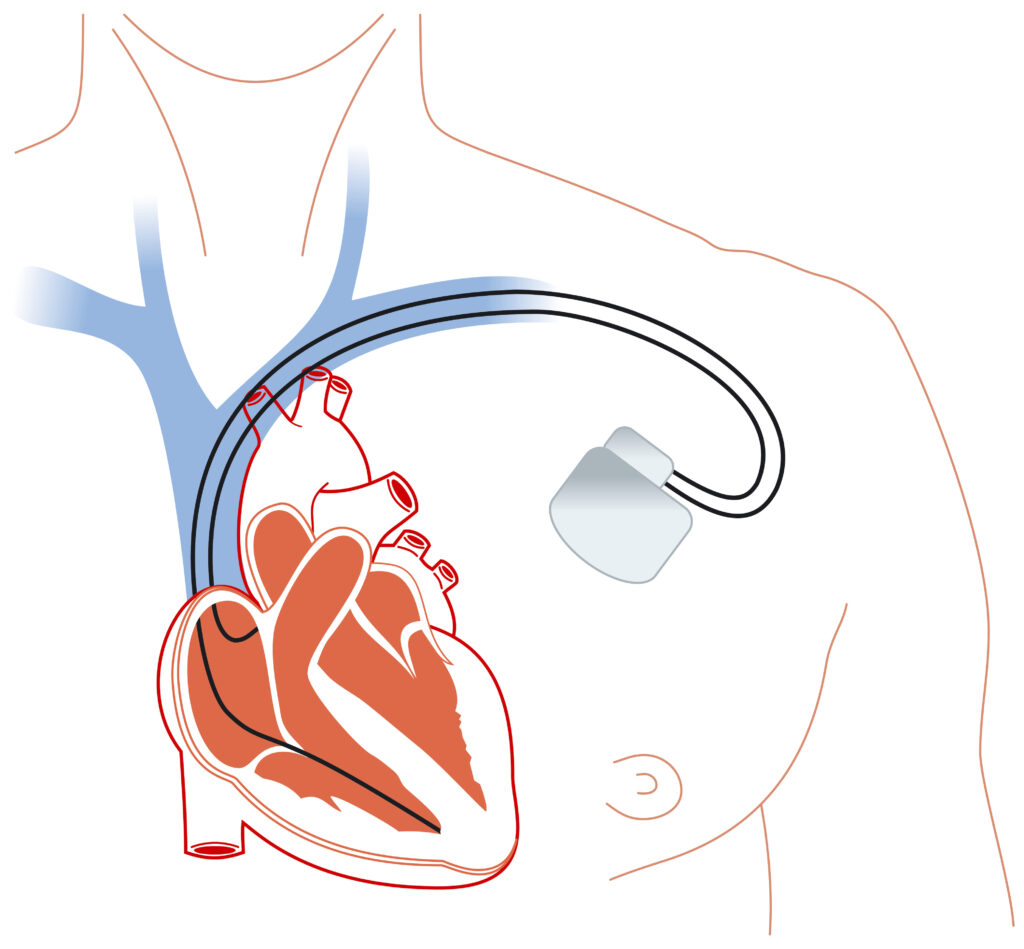
If you have an implanted defibrillator, a synthetic blood vessel, a heart patch, or a surgical conduit, a material used in some devices has been associated with gradual calcification in published studies (Journal of Vascular Surgery, Annals of Vascular Surgery).
The material is called ePTFE, short for expanded polytetrafluoroethylene. It has been widely used in medical devices for several decades. It has recently been referenced in connection with an FDA Class I recall involving specific defibrillator leads.
In July 2025, the FDA issued a Class I recall covering approximately 354,000 ePTFE-coated defibrillator leads after they were linked to 386 serious injuries and 16 patient deaths. Lawsuits are being filed. This article summarizes currently available information and research to help patients better understand the topic.
What Is ePTFE and Why Is It in Your Body?
ePTFE is a synthetic material: flexible, porous, and chemically stable. It is the medical-grade cousin of the material used in waterproof outdoor clothing (Gore-Tex), engineered for use inside the human body.
Surgeons and device makers chose ePTFE because, on paper, its properties looked ideal. It bends without breaking. It does not corrode. Its microscopic pores were supposed to let surrounding tissue grow into the device, helping it integrate with the body.
That choice put ePTFE almost everywhere in modern medicine. It is the insulating coating on the wires, called leads, that connect implantable defibrillators to the heart. It is the material in synthetic blood vessels, called grafts, used in bypass surgery, dialysis access, and vascular trauma repair. It is in the patches surgeons use to close holes in the heart, in the tubes used to redirect blood flow in children with single-ventricle heart defects, in the sutures used to repair leaky heart valves, and in some cosmetic facial implants.
The footprint is enormous. W.L. Gore alone has sold more than 600,000 ePTFE vascular grafts over the past decade and more than 755,000 cardiovascular patches over forty years. Several other manufacturers: Bard, LeMaitre, Atrium, Artivion, and B. Braun produce their own ePTFE grafts. Hundreds of thousands of ePTFE-coated defibrillator leads are still implanted in patients today.
The Calcification Problem
Here is the issue: information about long-term calcification has not always been consistently emphasized in publicly available patient-facing materials for some devices.
The same porous structure that lets tissue grow into ePTFE also lets minerals, proteins, and cell debris seep in. Over months and years, calcium phosphate crystals, the same compound your bones are made of, form inside those pores. The material stiffens. It loses flexibility. In some reported cases, this process has been associated with reduced device performance or failure.
This process is called dystrophic calcification. The timeline varies. Calcification has been documented as early as six months after surgery. More commonly, problems emerge years later. In many cases described in the literature, calcification may progress without noticeable symptoms until later stages.
What is most important to understand is that this is not a new discovery. The medical literature includes documented cases and studies on ePTFE calcification spanning several decades, though these findings are based on specific case reports and limited datasets:
- 1980s: The first reported case described a 28-year-old woman whose ePTFE dialysis graft occluded after only two years; on examination, it was filled with thrombus and a layer of calcified tissue.
- 2010: A published study found that calcium deposits were frequently present in failed PTFE grafts and were associated with thrombotic occlusion, graft disruption, and final graft failure.
- 2019: A clinical case series specifically identified Endotak Reliance defibrillator leads, and only Endotak Reliance leads, as developing shocking-coil calcification with abnormally high electrical impedance.
- 2020: A study showed that ePTFE grafts can begin showing calcification within just six months of implantation.
- 2023: A systematic review documented 73 reported cases of ePTFE graft calcification across a 35-year period.
- July 2025: Boston Scientific issued a physician advisory letter; the FDA followed with a Class I recall.
By the time this issue reached the public in 2025, peer-reviewed research had been describing it for over forty years.
Devices Affected
The recall that brought ePTFE into public view covers the Endotak Reliance family of defibrillator leads, the wires that connect an implantable cardioverter-defibrillator (ICD) to the heart muscle.
Three models are affected: ENDOTAK RELIANCE G, ENDOTAK RELIANCE SG, and ENDOTAK RELIANCE G/SG with 4SITE Connector. They were manufactured between 2002 and 2021. About 354,000 of these devices remain in service today.
The proposed mechanism, as described in published studies, involves gradual mineral accumulation within the material structure. The ePTFE coating on the leads shock coil, the part of the lead that delivers a life-saving electrical shock during cardiac arrest, slowly accumulates calcium. The calcium acts as an electrical insulator. Less of the shock energy reaches the heart muscle. The defibrillator may not be able to restart a stopped heart.
Clinically, this shows up as a rising “shocking impedance,” the resistance the electrical current has to push through to reach the heart. Boston Scientific now recommends that any lead with a shocking impedance above 150 ohms be considered for replacement.
As of July 2025, 386 serious injuries and 16 deaths have been reported in association with this issue. A 2026 analysis of FDA adverse-event reports found 2,378 events from January 2000 through September 2025; 54.9 percent of cases ultimately required lead replacement or extraction.
Removing a calcified lead is itself a high-risk procedure. In a published analysis of 11,304 lead extractions, major complications required urgent cardiac surgery 16 percent of the time, and 34 percent of those patients died.
Vascular Grafts
Vascular grafts are synthetic tubes that surgeons use to replace or bypass diseased blood vessels, most commonly in the legs of patients with peripheral artery disease, in the arms of patients on dialysis, and after vascular trauma.
W.L. Gore is the largest manufacturer, with more than 600,000 Gore-Tex grafts implanted over the past decade. Other manufacturers selling ePTFE vascular grafts include Bard (Carboflo, Distaflo, Dinaflo, Venaflo, IMPRA), LeMaitre (LifeSpan), Atrium (iCast, Advanta V12), Artivion, and B. Braun.
The mechanism is the same one. As calcium accumulates in the graft wall, the tube stiffens. Blood flow becomes turbulent. Clots form. In one of the published cases, a patient’s femoral artery bypass became “rigid and bony hard” nine years after the original surgery, and the entire graft had to be replaced under emergency conditions.
The contrast with newer alternatives is striking. In animal studies comparing tissue-engineered vascular grafts to ePTFE, the ePTFE grafts accumulated 269 calcium units per cubic centimeter on CT imaging. The tissue-engineered grafts accumulated 0.1. That is a 2,500-fold difference.
Pediatric Heart Conduits: the Fontan Procedure
Children born with single-ventricle heart defects, about 1 in every 2,000 live births, typically undergo a series of operations called the Fontan procedure. The final step usually involves implanting a Gore-Tex ring-supported PTFE conduit, 20 or 22 millimeters wide, to redirect venous blood directly to the lungs.
These conduits stay implanted for decades. The longer they remain, the more calcification can accumulate. A 2024 review in Pediatric Cardiology identified ectopic calcification as a leading long-term complication of synthetic materials in congenital heart surgery, leading to narrowing of the conduit, thrombosis, and eventual failure that often requires further open-heart surgery.
For families of Fontan patients, this is a difficult chapter. The decision to implant ePTFE was made when the child was a toddler. The consequences may not surface until adulthood.
Cardiovascular Patches and Mitral Valve Sutures
ePTFE is also used in surgical patches to close holes in the heart and reconstruct great vessels. The Gore-Tex Cardiovascular Patch alone has been used in more than 755,000 procedures over forty years.
In mitral valve repair, surgeons use Gore-Tex sutures known as CV-4 and CV-5 to replace the natural cords (chordae tendineae) that hold the valve leaflets in place. Over time, these sutures can calcify, weaken, and rupture, sending a patient back into surgery for a second valve repair. Published rupture rates in long-term follow-up are about 1.8 percent for the smaller CV-5 and 0.2 percent for the larger CV-4.
Why This Matters Now
The July 2025 FDA recall is what brought ePTFE calcification into public view (FDA Medical Device Recalls).
It is also worth noting that in December 2024, seven months before the FDA forced action on the Endotak Reliance leads, the FDA approved Humacyte’s SYMVESS, an acellular tissue-engineered vessel that is gradually replaced by the patient’s own tissue. In clinical studies, SYMVESS showed an amputation rate of 9.8 percent in vascular trauma patients, compared with 24.3 percent for synthetic grafts. The infection rate was 2.0 percent versus 8.4 percent. And as noted earlier, the calcium accumulation difference was a 2,500-fold reduction.
In other words, by the time the FDA acted on ePTFE leads in 2025, an alternative material with dramatically better outcomes already had FDA approval. These developments have raised questions among regulators, researchers, and legal professionals about long-term risk communication and device design.
Step 1: Check If You’re Affected
There are two questions to answer.
Do You Have an ePTFE Device?
Most patients never see their device’s specifications. Here is how to find out:
- Find your implant card. When you received your device, you should have been given a wallet-sized card listing the manufacturer, model number, and serial number. If you have it, look for any of these brand names: Endotak Reliance, Gore-Tex, GORE, ePTFE, expanded polytetrafluoroethylene, PTFE, IMPRA, Advanta, Carboflo, Distaflo, Dinaflo, Venaflo, LifeSpan, iCast, Bard Composix, or SoftForm.
- Request your operative report. If you do not have your card, call your surgeon’s office or the medical records department of the hospital where you had surgery. The operative report will name the manufacturer and product code of any implanted device.
- Use the manufacturer’s lookup tool. If you have a Boston Scientific cardiac device, you can look up your device specifications at bostonscientific.com → Patient Resources → Device Lookup Tool, or call customer service.
Are There Warning Signs?
Calcification often progresses silently. By the time symptoms appear, the device may be close to failure. Watch for the following:
- The defibrillator leads patients. Ask your cardiologist for your most recent shocking impedance reading. Above 150 ohms is the published replacement threshold. Watch for unexpected shocks, device alerts, palpitations during exertion, or unexplained fainting.
- Vascular graft patients. Sudden limb pain, coldness, or color change is a vascular emergency. Other warning signs include a hard or pulsating mass at the graft site or non-healing wounds near a dialysis access.
- Fontan or pediatric heart patients. New shortness of breath, decreased exercise tolerance, or new arrhythmias years after surgery warrant a cardiac MRI or CT to check for conduit calcification.
- Mitral valve repair patients. New or worsening regurgitation symptoms: fatigue, breathlessness with activity, could indicate suture rupture.
The general rule: if you have any ePTFE device, ask your physician whether monitoring should be intensified given the July 2025 FDA recall and the published calcification evidence.
Step 2: Contact Justpoint PLLC
If your check raises concerns, the next step is a free, no-obligation case evaluation with Justpoint PLLC. The consultation is confidential. There is no cost. Consulting with us does not create an attorney-client relationship until a written agreement is signed by both parties.
Why does timing matter? Each state has its own statute of limitations, the deadline for filing a claim. The “discovery rule,” which delays the clock until you knew or reasonably should have known the device caused harm, is recognized in only about a dozen states. Some states also impose statutes of repose: absolute deadlines, often 10 to 12 years from when the device was sold, that bar a claim regardless of when the injury came to light. Deadlines may vary depending on jurisdiction and individual circumstances, so patients may wish to seek legal advice if they have concerns.
Litigation context. ePTFE-coated medical devices have an established product liability track record. C.R. Bard reached a $184 million settlement for Kugel hernia mesh claims in 2011. A broader Bard/Davol settlement wave covering approximately 38,000 cases was anticipated to total roughly $1.7 billion in 2024-2025. The Atrium C-Qur multi-district litigation settlement plan was approved by a federal judge in October 2024. The Endotak Reliance situation sits squarely within this same pattern.
If you or a loved one has been harmed by an ePTFE-coated medical device, you may be entitled to compensation.
Individuals with concerns about potential harm may consider seeking independent legal advice to better understand their options.
Contact Us
This article was published by Justpoint Inc. The information provided is for educational purposes, does not constitute medical or legal advice and is based on publicly available research and regulatory communications. Findings may vary depending on individual circumstances, device type, and clinical context. If you have a medical device that may be affected, please consult your physician. If you believe you may have a legal claim, please consult a qualified attorney.
Justpoint Inc. supports Justpoint Law (Justpoint PLLC) by providing operational and administrative services. All legal services are provided by Justpoint Law, and any client-related communications are made on its behalf.
